Published on February 26th 2026 | 10 mins , 1980 words
Table 1 shows the periodic table of elements
(a) Explain each of the following:
(i) Atomic radius of the elements decreases from magnesium to chlorine. (1 mark)
Because the nuclear charge increases from magnesium to chlorine due to the increasing number of protons.
(ii) Ionization energy of the elements increases from magnesium to chlorine.(1 mark)
Because of the decrease in atomic radius hence increase the nuclear charge.
(b) Explain each of the following in terms of structure and bonding.
(i) The melting point of aluminium is higher than that of magnesium. (1 mark)
Both Mg and Al are metals a giant metallic structure; Al has stronger metallic bond because it has 3 delocalised electrons hence require more energy to break metallic bond.
(ii) The melting point of phosphorus is higher than that of chlorine. (1 mark)
\(P_{4}\) and \(Cl_{2}\) exist as simple molecular structures held by weak Van der waals forces. \(P_{4}\) molecules are much larger and heavier than \(Cl_{2}\) molecule hence has stronger Van der waals forces than \(Cl_{2}\)
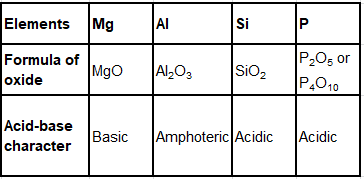
(c) Complete Table 2 by giving the formula of the oxides of the elements and their acid-base character.
(i) Name the type of bond in the chloride. (1 mark)
(ii) Determine the oxidation number of sulphur in the chloride. (1 mark)
\(2x+2(−1)=0 \)
\(2x−2=0\)
\(2x=+2 \)
\(x=+1\)
(e) Silicon tetrachloride fumes in air.
Write an equation for the reaction. (1 mark)
\(SiCl_{4}\) (l)+ \(2H_{2}O(g)\)⟶\(SiO_{2}(s)\)+ \(4HCl(g)\)
(f) Silicon is a metalloid.
(i) State what is meant by the term metalloid. (1 mark)
Is an element that exhibits properties of both metals and non-metals.
(ii) State one use of silicon as a metalloid. (1 mark)
— Used in manufacture of electronic components.
— Used in making porcelain and refractory materials.
(a) State the names of the following compounds.
(i) \(CH_{3}(CH_{2})_{5}CH_{3}\) (1 mark)
Heptane.
(ii)\( CH_{3}(CH_{2})_{3}C≡CH\) (1 mark)
Hex-1-yne.
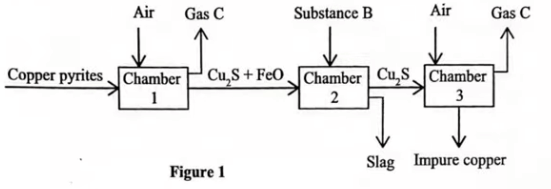
(b) Figure 1 shows extraction of copper from copper pyrites, \(CuFeS_{2}\).
(a) Identify gas C. (1 mark)
Sulphur (IV) Oxide // \(SO_{2}\).
(b) Write an equation for the reaction that takes place in chamber 1. (1 mark)
\(2CuFeS_{2}\)(s)+\(4O_{2}\)(g)⟶\(Cu_{2}\)S(s)+2FeO(s)+\(3SO_{2}\)(g)
(c) Identify substance B. (1 mark)
Silicon (IV) Oxide // \(SiO_{2}\)
(d) Give the formula of the main compound in the slag. (1 mark)
\(FeSiO_{3}\).
Write an equation for the two reactions that take place in chamber 3
\(2Cu_{2}S(l)\) + \(3O_{2}(g)\) ⟶ \(2Cu_{2}O(l)\) + \(2SO_{2}(g)\)
\(Cu_{2}S(l)\) + \(2Cu_{2}O(l)\)⟶ \(6Cu(l)\) + \(SO_{2}(g)\)
(f) Describe the process that is used to purify the impure copper. (2 marks)
Impure copper is purified by using electrolysis. The impure is used as anode and a thin sheet of pure copper is used as the cathode. An aqueous solution of copper (II) sulphate is used as the electrolyte. When current is passed, copper dissolves from the impure anode and it is deposited as pure copper on the cathode.
(g) When left exposed for along time in air, copper coins turn green. Explain this observation. (1 mark)
Copper reacts slowly with Oxygen, Carbon (IV) oxide and water vapour in the air to form a green coating of basic copper (II) carbonate.
3 (a) State what is meant by standard enthalpy of formation of a compound. (1 mark)
It is the enthalpy change when one mole of a compound is formed from its constituent elements in their standard states under standard conditions.
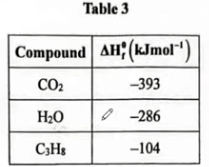
(b) Table 3 gives enthalpy of formation of some compounds.
(i) Write the thermochemical equations for the formation of:
I. \(CO_{2}\) ; (1 mark)
C(s)+ \(O_{2}\)(g)⟶ \(CO_{2}\) (g) \(ΔHf^{θ}\)=−393 \(kJmol^−1\)
II. \(H_{2}O\); (1 mark)
\(H_{2}(g)\) + \(\frac{1}{2}O_{2}(g)\) ⟶ \(H_{2}O(l)ΔHf^{θ}\) =−286 \(kJmol^{−1}\)
(ii) Study the energy level diagram in Figure 2 and answer the questions using the data in Table 3.
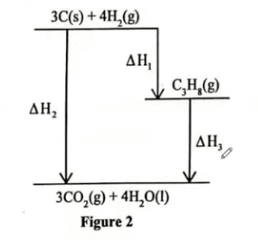
\(ΔH_{1}\) =−104 \(kJ mol^{−1}\)
II. Calculate the values of:
\(ΔH_{2}\) . (2 marks)
\((ΔH)\) = \([3×ΔHf∘(CO2)]+[4×ΔHf∘(H_{2}O)] \)
\(=[3×(−393)]+[4×(−286)] kJ \)
\(=(−1179)+(−1144) =−2323 kJ mol^{−1}\)
\(ΔH_{3}\). (1 mark)
\(ΔH_{2}\) = \(ΔH_{1}\) + \(ΔH_{3}\)
\(ΔH_{3}\)= \(ΔH_{2}\) − \(ΔH_{1}\)
=−2323+104 = \(−2219 kJ mol^{−1}\)
(iii) When a sample of butane \((C_{4}H_{10})\) was burned, the heat produced raised the temperature of 500 g of water by 35°C.
(Specific heat capacity of water = \(4.2 J g^{−1}K^{−1})\)
Calculate the:
I. heat change for the reaction. (1 mark)
\(Q=500g×4.2 J g^{−1}K^{−1}×35K\)
=73,500 J
=73.5 kJ
II. mass of butane that was burned given that the heat of combustion of one mole of butane is \(−2880 kJ mol^{−1}\) . (C=12.0,H=1.0) (2 marks)
n= \(\frac{73.5 kJ }{ 2880 kJ/mol}\)
=0.02552 moles
Mass=0.02552 moles×58.0 g/mol
=1.48 g
4. (a) Describe how a sample of chlorine gas can be prepared using the following compounds: solid sodium chloride, solid manganese (IV) oxide and concentrated sulphuric (VI) acid. (3 marks)
Mixture a suitable amount of solid NaCl and solid \(MnO_{2}\) in a round bottomed flask.
Add concentrated \(H_{2}SO_{4}\) to the mix. Heat the mixture gently.
Pass the gas through water then through concentrated \(H_{2}SO_{4}\) . Collect the gas downward delivery.
(Note: Accept diagram/setup)
(b) Figure 3 is a flowchart showing some reactions of chlorine.
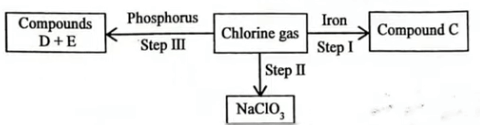
(i) Give the name of compound C formed in step I. (1 mark)
Iron (III) chloride
(ii) State the reagent and conditions necessary for the reaction in step II. (1 mark)
Reagent:
Hot concentrated sodium hydroxide
Conditions: High temperature
(iii) Two compounds are formed in the reaction in step III. Give the formulae of each of the two compounds. (2 marks)
I. D; \(PCl_{3}\)
II. E; \(PCL_5\)
(c) Write an equation for the reaction that takes place when chlorine gas reacts with aqueous hydrogen sulphide. (1 mark)
\(Cl_{2}\)(g)+\(H_{2}S\) (aq)→2HCl(aq)+S(s)
(d) State the observation made when:
(i) chlorine gas is bubbled into aqueous potassium iodide. (1 mark)
The solution changes from colourless to brown.
(ii) moist red-litmus paper is placed in a gas jar of chlorine gas. (1 mark)
The red litmus paper turns first red then white (bleached)
(e) Write an ionic equation for the reaction that takes place when aqueous lead (II) nitrate is added to aqueous potassium chloride. (1 mark)
\(Pb^{2+}(aq)\) + \(2Cl^{-}(aq)\) → \(PbCl_{2}(s)\)
(f) State the reason hy chlorine is used in water treatment. (1 mark)
It is an effective disinfectant or germicide.
(a) Figure 5 shows the Down's cell used in the extraction of sodium from molten sodium chloride.
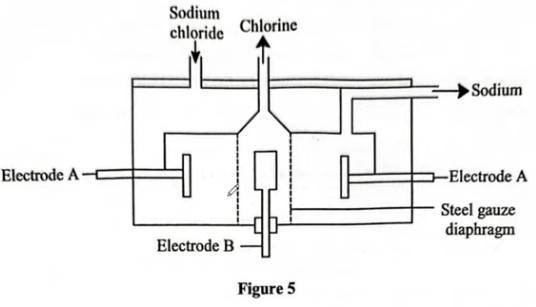
Anode: B
Reason: Chloride ions are discharged to form chloride gas.
Cathode: A
Reason: Sodium ions are discharged to form molten sodium.
(ii) State the purpose of the steel gauze diaphragm. (1 mark) It prevents recombination of sodium at the cathode and chlorine gas at the anode to NaCl.
(b) Table 4 shows the melting point and boiling point of sodium and sodium chloride.
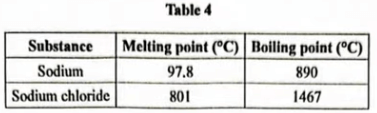
By adding calcium chloride to the sodium chloride.
(ii) State the advantage of operating at 600°C. (1 mark)
It save the cost of electricity used in heating since is economical.
(iii) State the physical state in which sodium is obtained from the cell. Give a reason. (2 marks)
Liquid/Molten; The operating temperature of the cell (600°C) is above the melting point of sodium.
(c) Starting with 1000 kg of sodium chloride, calculate the mass of sodium produced. (Na=23.0;Cl=35.5) (2 marks)
Mass=\(\frac{1000 kg×23}{ 58.5 }\)
=393.16 kg
(d) Explain how a sample of solid sodium should be stored in the laboratory. (2 marks)
It should be stored under paraffin; This prevents the sodium from coming into contact with air or moisture being that it is very reactive.
(e) State one industrial use of sodium. (1 mark)
- As a reducing agent in the extraction of metals like Titanium from its chloride.
- Used in sodium vapour lamps to give yellow light.
- An alloy of K and Na is used as a coolant.
6. The products of electrolysis depend on the conditions used.
(a) (i) State the conditions, if the products of electrolysis of aqueous copper (II) sulphate are:
I. oxygen and copper; (1 mark)
Using inert electrodes.
II. copper. (1 mark)
Using a copper electrode.
(ii) During electrolysis of an electrolyte containing aqueous \(A^{3+}\) ions, 19.4 g of A were obtained when a current of 15 amperes was passed through it for 2 hours.
I. Write the half equation for the reaction that took place. (1 mark)
\(A^{ 3+ }(aq)\) +\(3e^−\)→A(s)
II. Determine the relative atomic mass of A. (2 marks) (1 Faraday = 96, 500 coulombs)
Q=15A×7200s =108,000C
RAM=\(\frac{108000×19.4}{ 96500 \times 3}\)
=52.0
(b) Study the following cell representation diagram and answer the questions that follow.
\(Ni(s)/Ni^{2+}\)(aq)// \(Ag^{+}\) (aq)/Ag(s) \(E_{cell}^{θ}\)=+1.05V
(i) State what the symbol // represents in the diagram. (1 mark)
Salt bridge
(ii) Write an equation for the reaction that occurs in the cell. (1 mark)
Ni(s)+\(2Ag^{+}(aq)\) →\(Ni^{ 2+}\) (aq)+ 2Ag(s)
(iii) Given that the \(E^{θ}\) of \(Ag^{+}\) (aq)/ \(Ag(s)\) half-cell is +0.80V, calculate the \(E^{θ}\) of the \(Ni^{2+}(aq)/Ni(s)\) half-cell. (1 mark)
(iii) Given that the Eθ of Ag+(aq)/Ag(s) half-cell is +0.80V, calculate the Eθ of the Ni2+(aq)/Ni(s) half-cell. (1 mark)
\(E^{θ}_{Ni^{2+}/Ni}\) = \(E^{θ}_{Ag+/Ag}\) − \(E^{θ}_{cell}\)
= +0.80V−1.05V
= −0.25V
(iv) State how e.m.f of a cell can be used to predict whether a reaction is feasible. (1 mark)
A reaction is feasible if the electromotive force for that reaction is positive.
(c) Corrosion of a metal involves formation of an oxide layer on the metal surface. Explain why corrosion of aluminium is protective while that of iron is destructive. (2 marks)
When Al corrodes, it forms aluminium oxide on its surface that prevent further reaction hence protective. When Fe corrodes, it forms a rust. Rust is flaky; this exposes new metal to the environment, allowing corrosion to continue until the entire object is consumed. Thus it is destructive.
7. (a) State and explain how the rate of the reaction A(g)+B(g)→AB(g) is affected by an increase in:
(i) pressure; (2 marks)
An increase in pressure decreases the volume occupied by gas molecules thus increases the concentration of the reactants hence increasing the rate of effective collisions. This results into an increase in the rate of reactions.
(ii) temperature. (2 marks)
An increase in temperature increases the kinetic energy of the reacting particles by increasing their collision frequency. Increase of kinetic energy provides the reacting particles with necessary activation energy for the reaction to occur. The reaction therefore proceeds at a faster rate.
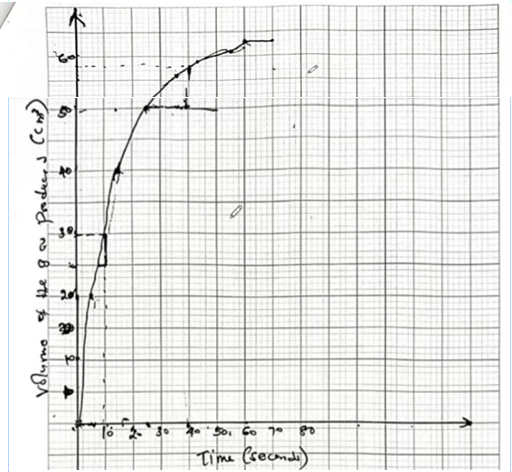
(b) The rate of reaction between ethanoic acid and copper (II) carbonate can be monitored by measuring the volume of gas produced. The results obtained using 75.0 cm3 of 0.1 M ethanoic acid and excess copper (II) carbonate are shown in Table 5.
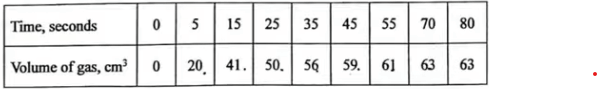
\(2CH_{3}COOH(aq)\) + \(CuCO_3\) (s)→ \(Cu(CH_{3}COO)_{2}(aq)\) + \(CO_{2}\) (g)+ \(H_{2}O(l)\)
(ii) Give a reason why excess copper (II) carbonate was used. (1 mark)
To ensure that \(CH_{3}COOH\) is the limiting reactant.
To ensure that the reaction goes to completion until all the acid is used up.
(iii) On the grid provided, draw a graph of the results obtained. (3 marks)
(iv) Use the graph to determine the rate of reaction at: I. 10 seconds; (1 mark)
\(\frac{30−25}{ 10−6 }\)
=5/4
= \(1.25 cm^{3}/s\)
II. 40 seconds. (1 mark)
\(\frac{58−50}{ 40−25 }\)
=8/15
≈ \(0.53 cm^{3}/s\)
(v) Give a reason for the difference between the two average rates calculated in (iv). (1 mark)
The rate of reaction decreases from 10s to 40s because the concentration of limiting reactant decreases over time, resulting in a lower frequency of effective collisions.